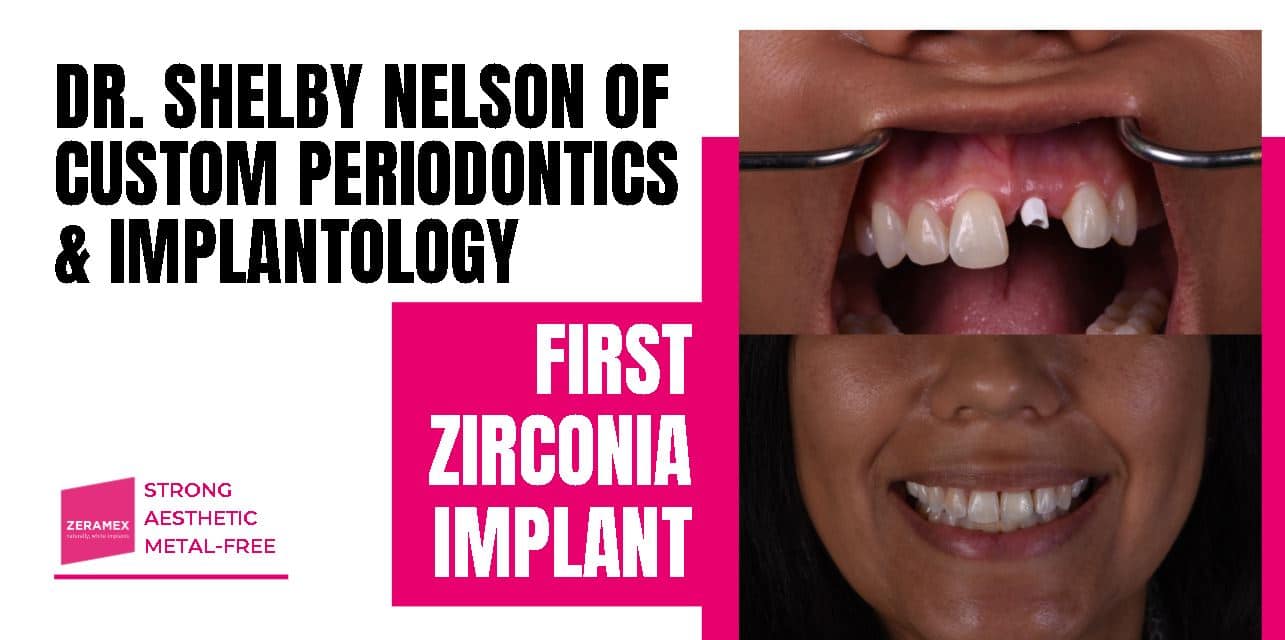
Dr. Shelby Nelson of Custom Periodontics & Implantology shares her experience in placing her first ever Ceramic Dental Implant. Continue reading to know the reasons why she chose to move forward with ZERAMEX XT!
ZERAMEX XT is featured in Inside Dentistry. Case Presentation featuring Zeramex Ceramic Dental Implants by Paresh Patel, DDS | Apolinar Madrigal, DDS
ZERAMEX XT is on the front page of Implant Practice – US featuring Dr. Paresh Patel’s experience with ZERAMEX Ceramic Dental Systems
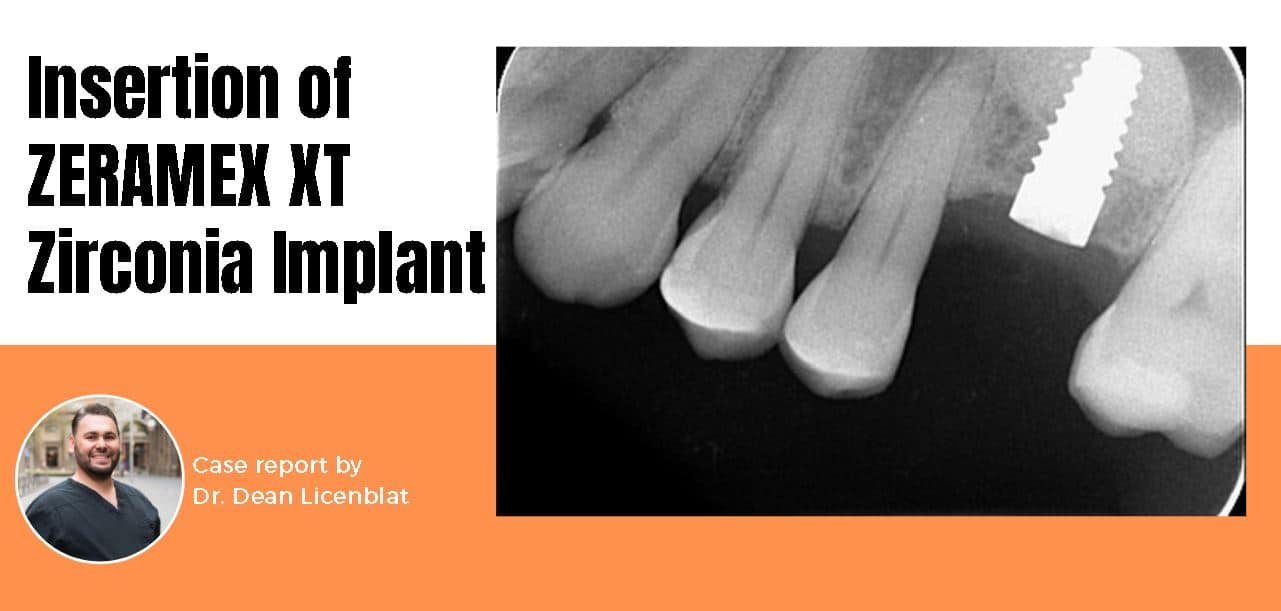
A social media case presentation by Dr. Dean Licenblat | Posted on Instagram last November 26, 2020 | Zirconia implants are another way of solving the problem of edentulism.
This case report demonstrates the straightforward surgical and restorative protocol for the ZERAMEX XT Implant. This treatment option has proven valuable in cases where the doctor wishes to provide the optimal esthetic solution, particularly in anterior cases with thin gingival biotype, where titanium implants often produce a gray shadowing.
Social Media Case Report posted on LinkedIn by Dr. Luis Alicea – Your Desired Smile – All on-8 on the upper arch & All on-6 on the lower arch with guided ceramic implants
Social Media Case Report posted on Facebook by Dr. Luis Alicea – Your Desired Smile – Seven guided ZERAMEX Zirconia Implants placed
If you watched Dr. GeorgiTopic: Zirconia ceramic implants: a viable alternative in dental implantology.
Hot Seat part 17- Dr. Omid Moghaddas with Dr. Sammy Noumbissi
If you watched Dr. Georgios Kotsakis’ lecture during the Pikos Symposium 2020 event last October 15-17 and you feel the need to to re-watch it; check out this video where Dr. Kotsakis goes in the hot-seat with Dr. Omid Moghaddas.
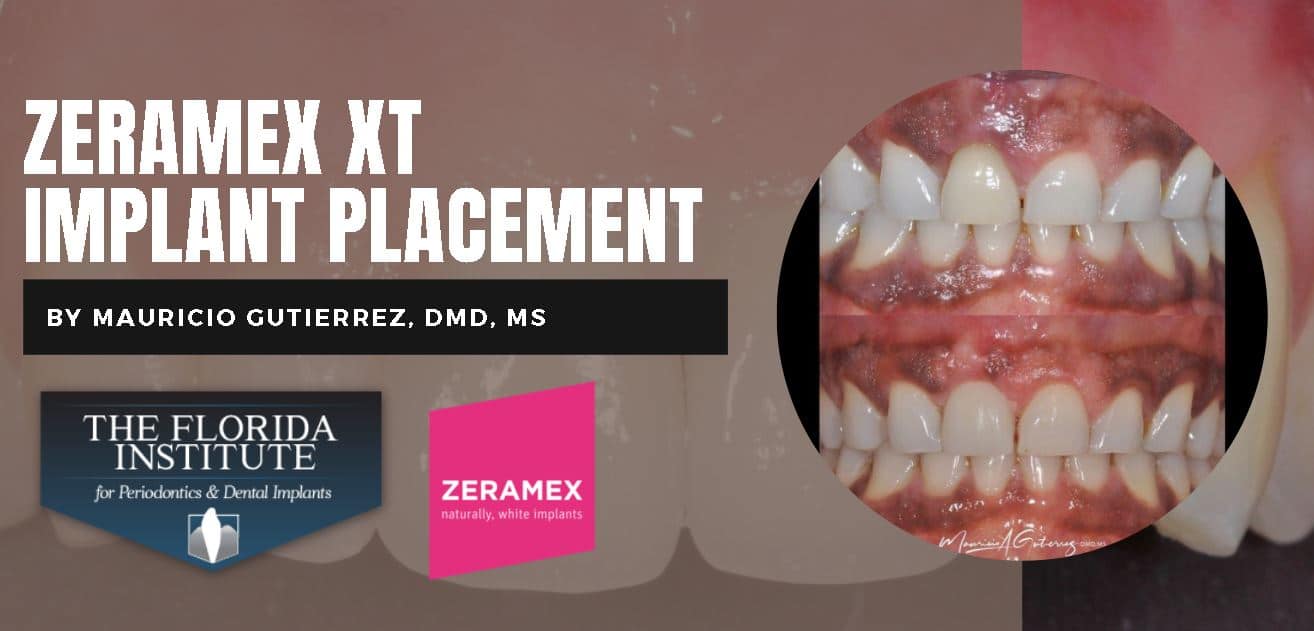
Check out this Social Media Case by Dr. Mauricio Gutierrez, DMD, MS | Florida Institute for Periodontics and Dental Implants featuring ZERAMEXT XT Ceramic Dental Implant