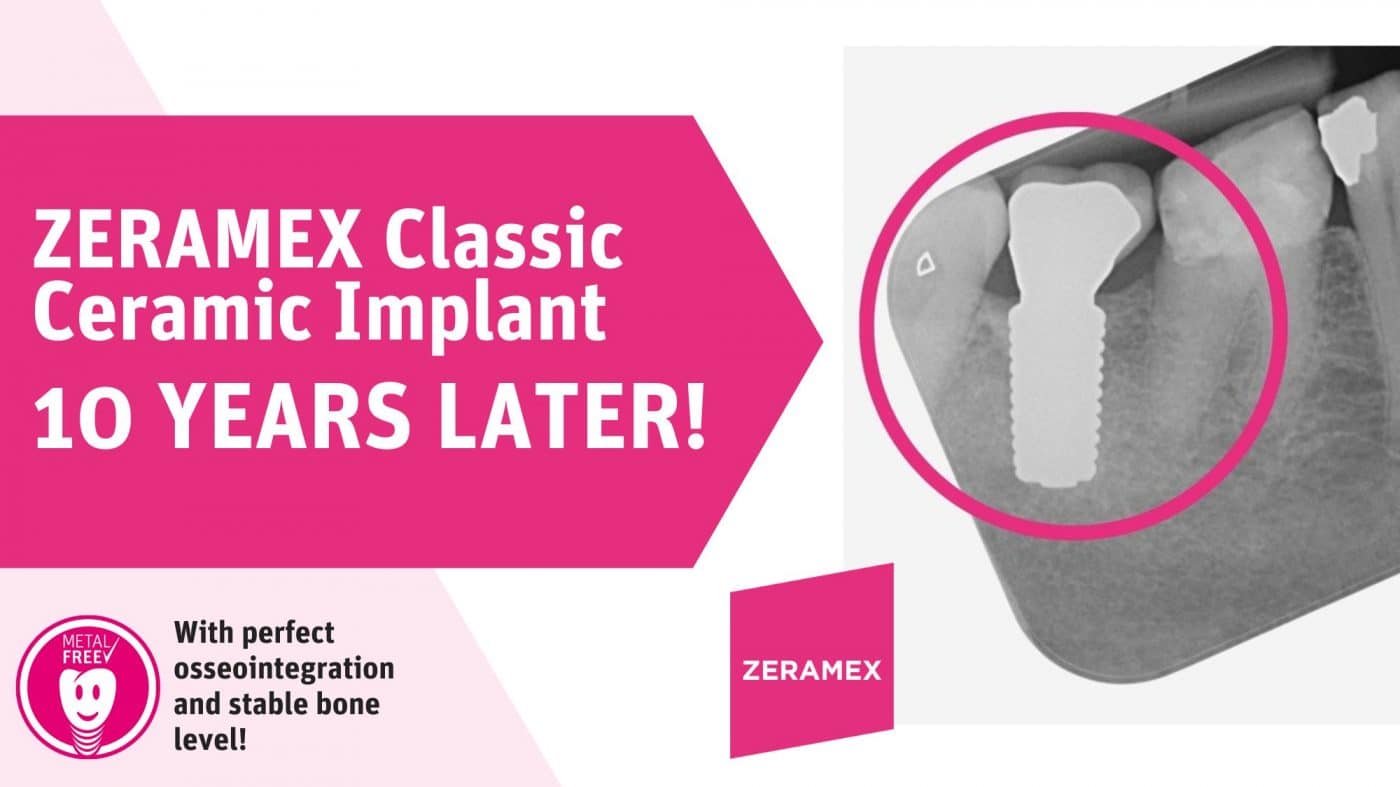
Source: ZERAMEX
This image is an X-RAY taken of one of the first ZERAMEX Implants ever placed and the implant still looks perfect!
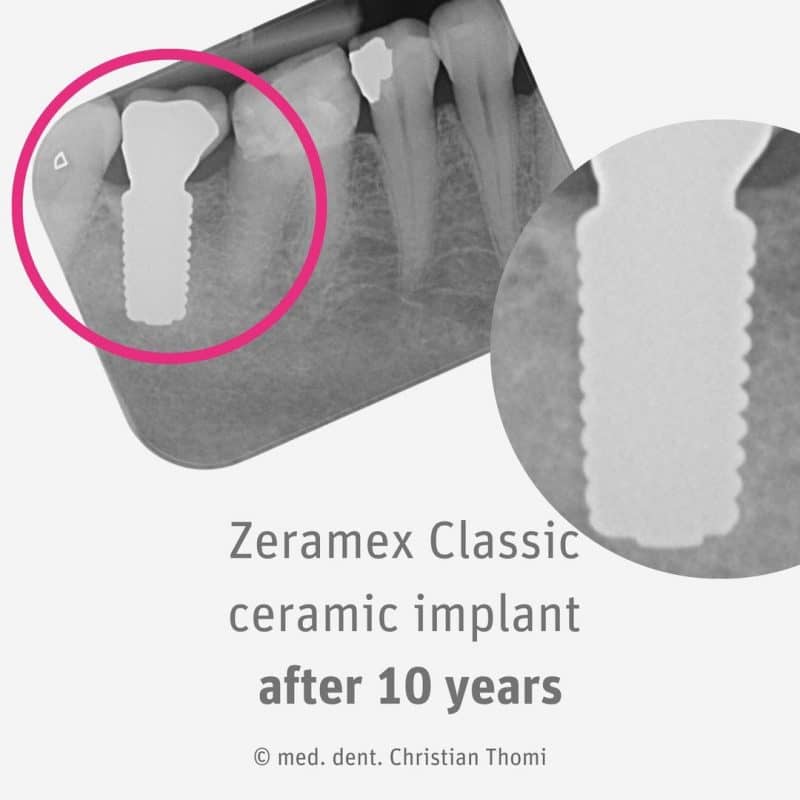
How Long Should My ZIRCONIA BioCeramic Dental Implant last?
The demand for metal-free alternatives in dental implants has increased. Patients are seeking alternatives to titanium dental implants for two main reasons: (1) The belief they suffer from a metal allergy. (2) They are seeking a holistic approach to their treatment (also referred to as biologic dentistry).
Titanium dental implants can provoke type I and IV hypersensitivity reactions that resolve only with the removal of the implant.1 Additionally, titanium corrosion and particle release has been linked to changes in the DNA of peri-implant microorganisms, increased risk and incidence of inflammatory disease and peri-implantitis.2
Zirconia has been shown to have equal capacity for osseointegration when compared to titanium3, and ability to withstand occlusal forces.4,5 Investigation periods of up to five years have reported survival and success rates of more than 95% in humans 6-8, making them a viable alternative for tooth replacement.
Patient demand for metal-free alternatives have increased significantly in the past five years and expected trends in global ceramic implant market shares predict that zirconia implants will capture about 10% of the market by 2025.
ZERAMEX Classic
The ZERAMEX Classic is a predecessor model of Zeramex XT. Despite being introduced more than 10 years ago, it continues to demonstrate excellent osseointegration and stable bone levels. It is a groundbreaking product in the field of metal-free implantology. It has proven to be a reliable and effective solution for patients looking for a stable, long-lasting dental implant.
One of the key benefits of the ZERAMEX Classic is its ability to achieve perfect osseointegration. This refers to the process by which the implant fuses with the surrounding bone tissue, providing a secure foundation for the replacement tooth. This is crucial for the long-term success of the implant and ensures that it stays in place for years to come.
In addition to its excellent osseointegration, it also boasts a stable bone level. This means that the implant does not cause any bone loss or deterioration over time, ensuring that the implant remains stable and secure.
Overall, the ZERAMEX Classic is a highly respected and trusted product in the field of metal-free implantology. Its ability to achieve perfect osseointegration and stable bone level makes it an excellent choice for patients looking for a reliable, long-lasting dental implant.
Founded in 2005, Dentalpoint sets future-orientated standards in dentistry.
The ZERAMEX® registered trademark stands for an aesthetic and 100% metal-free all-ceramic implant system that addresses a growing patient need.
Dentalpoint AG is the global market leader in the field of metal-free, two-part ceramic implants. With the development of a breakthrough production technology that significantly reduces production costs and machining time while allowing greater design freedom, Dentalpoint is the only company able to produce the high-quality titanium implant designs by leading manufacturers in ceramics.
In 2009, after four years of intensive research, the dental implant manufacturer presented the first convincing two-part implant system made of white high-performance ceramics, particularly with regard to the aesthetics and biocompatibility of the implants. The material, zirconium dioxide, which very closely resembles the natural tooth root and also satisfies the highest aesthetic demands is another innovation.
The ZERAMEX XT is the leading two-piece Zirconia, BioCeramic dental implant solution in the world.
Reversible screw-retained and 100% metal-free: The Zeramex XT boasts maximum prosthetic flexibility, thanks to an unlimited range and digital workflow.
The Zeramex XT ceramic implant is manufactured from hard zirconium oxide ATZ blanks manufactured by hot isostatic pressing. No thermal process (sintering) or post-processing takes place after the final shaping of the outer and inner geometry of the implant. This ensures that no further changes can occur in the material structure. This manufacturing process is very complex, requires a great deal of experience and expertise and enables a high shaping accuracy and precision in the manufacture of the implant.
The core of the connection is the Vicarbo screw.
VICARBO® is carbon-fiber reinforced, in which the carbon fibers are aligned with the longitudinal axis of the material. In this way, we can achieve enormous strength. Thanks to the production process developed by ZERAMEX®, the carbon fibers are not damaged during production and they retain their full function. This screw, for which a patent is pending, is unique in dental implantology!
The VICARBO® screw is a precision screw to optimally capture occlusal forces. When tightened, it grips the existing contour of the thread thanks to the significant different hardness of the ceramic and screw.
References
- Sicilia A, Cuesta S, Coma G, Arregui I, Guisasola C, Ruiz E, Maestro A. Titanium allergy in dental implant patients: a clinical study on 1500 consecutive patients. Clinical Oral Implants Research. 2008 Aug;19(8):823-35.
- Noumbissi S, Scarano A, Gupta S. A Literature Review Study on Atomic Ions Dissolution of Titanium and Its Alloys in Implant Dentistry. 2019 Jan 24;12(3):368. doi: 10.3390/ma12030368.
- Gahlert M, Rohling S, Wieland M, Sprecher CM, Kniha H, Milz S. Osseointegration of zirconia and titanium dental implants: a histological and histomorphometrical study in the maxilla of pigs. Clinical Oral Implants Research. 2009 Nov;20(11):1247-53.
- Andreiotelli M, Kohal RJ. Fracture strength of zirconia implants after artificial aging. Clinically Implant Dentistry and Related Research. 2009 Jun;11(2):158-66.
- Silva NR, Coelho PG, Fernandes CA, Navarro JM, Dias RA, Thompson VP. Reliability of one-piece ceramic implant. Journal of Biomedical Materials Research Part B: Applied Biomaterials. 2009 Feb;88(2):419-26.
- Grassi FR, Capogreco M, Consonni D, Bilardi G, Buti J, Kalemaj Z. Immediate occlusal loading of one-piece zirconia implants: five-year radiographic and clinical evaluation. International Journal of Oral Maxillofacial Implants. 2015 May-Jun;30(3):671-80.
Contact
Want to know more about Zeramex?
Email us at mfoley@zeramexusa.com with your contact details and we will get in touch with you!
ZERAMEX BROCHURE
Click here to download the Zeramex sales brochure