Aesthetic Transformations Utilizing Zirconia Implants
Written by: Paresh Patel, DDS | August 23, 2022 | Dentistry Today
Introduction
With new emerging technology, ceramic implants are some of the most innovative products that provide an alternative to traditional titanium implants. They have been shown to be strong, aesthetic, and peri-implant-tissue friendly, as well as a metal-free option. Some of the modern ceramic implant systems, such as Zeramex XT (Dentalpoint AG), allow for a metal-free solution for long-term success. This offers a choice to those patients who have a titanium sensitivity or otherwise wish to use a nonmetal solution. 1-3 In the cosmetic zone (maxillary anterior region), placing any type of implant can be challenging, especially with adjacent natural teeth.
Zirconia offers an inherent white color to mask any shimmer or show-through, making a “gray” shadow a thing of the past. Thin biotype tissue is especially a characteristic that has always been an aesthetic risk and can be minimized with this innovative implant system and its natural tooth color. Zirconia has also been shown to provide optimal blood circulation around the implant body, thus increasing the long-term stability of the perio-implant complex.
The following clinical case demonstrates the use of the Zeramex 2-piece ceramic implant that uses a unique carbon fiber fixation screw to offer a true metal-free solution for the aesthetic zone.
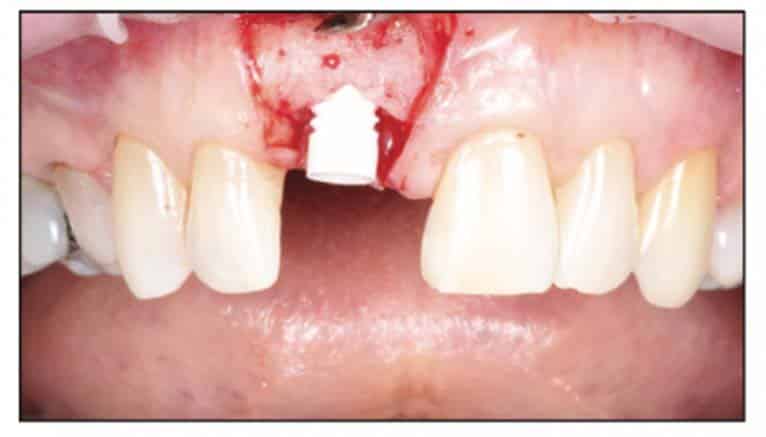
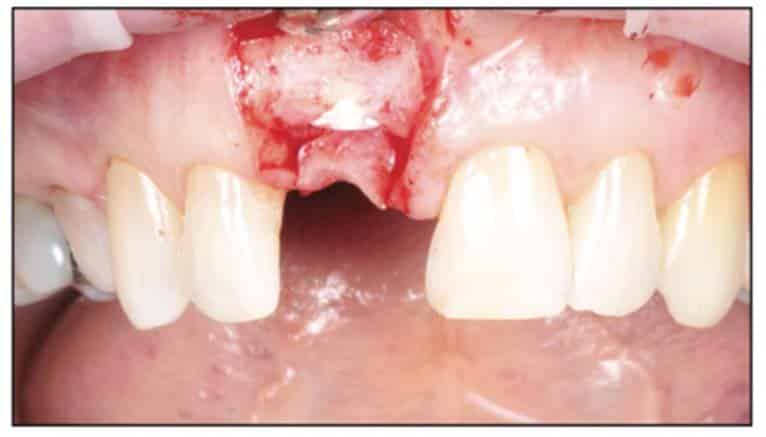
A 54-year-old female presented to our office to learn about some options to replace failing partial RCT on her upper right maxillary central incisor (Figure 1). Tooth No. 8 was extracted using W&H piezo EXT 1 and EXT 2 tips to reduce the risk of a buccal fracture. The site was preserved with a cortico-cancellous allograft (Newport Surgical) and allowed to heal for 5 months (Figure 2). After healing, smile photos were taken to assess the overall aesthetic risk of the case. Treatment options were discussed with the patient. With a ridge width of less than 6.5 mm, the patient decided to use a ceramic implant to minimize the potential for gray show-through. We proposed doing a veneer graft as well as a connective tissue graft to minimize these risks if a titanium implant were placed. The patient declined these surgical interventions (Figures 3 and 4).
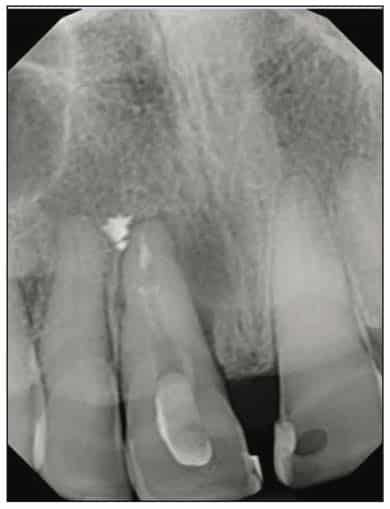
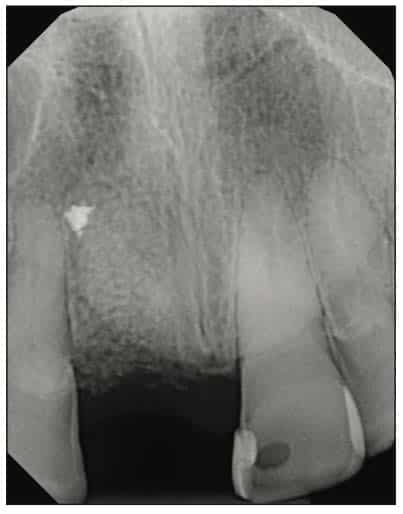
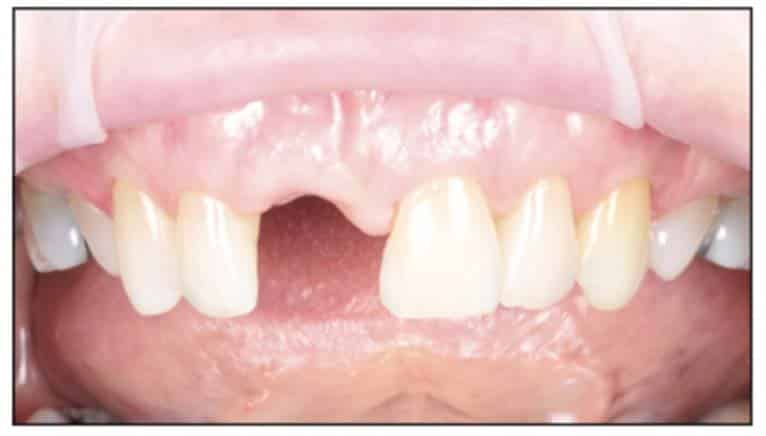
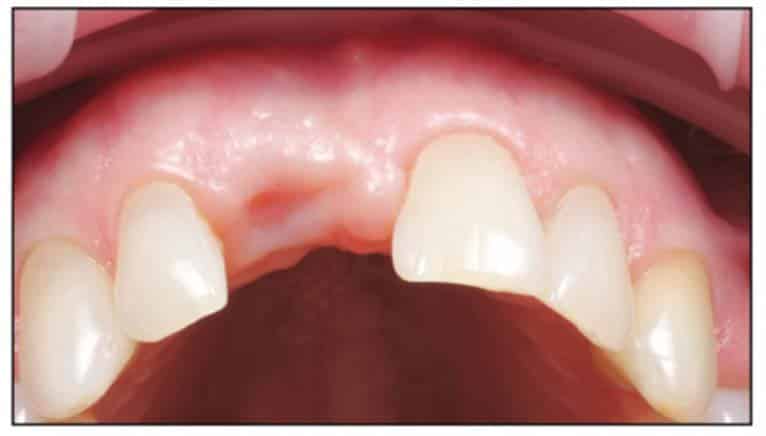
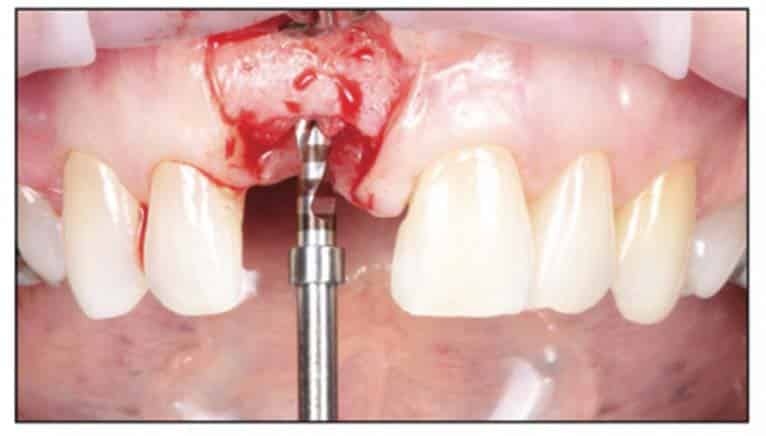
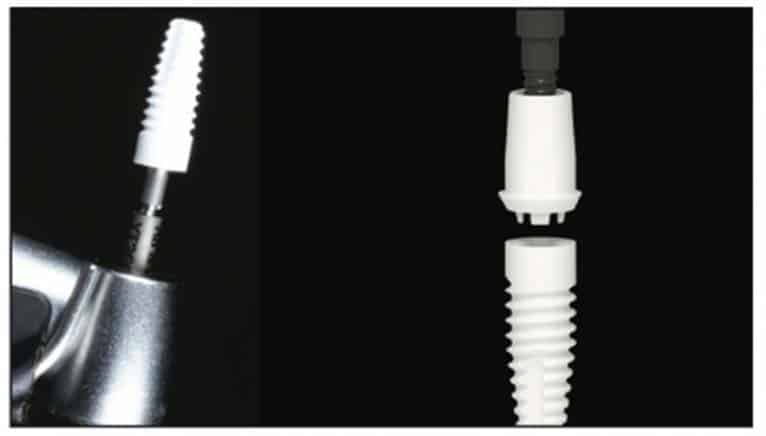
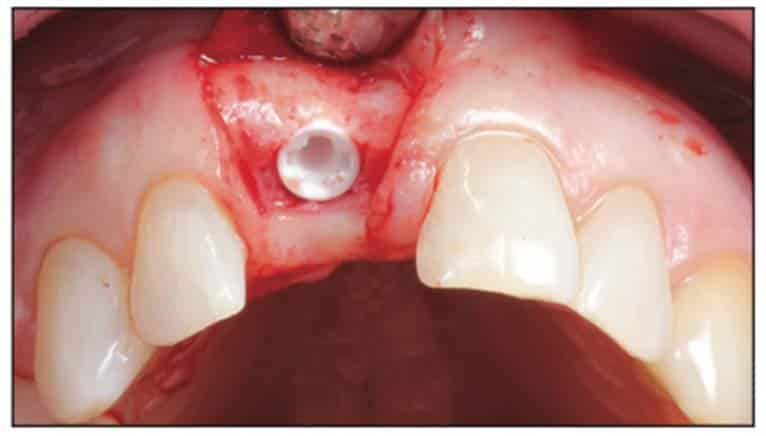
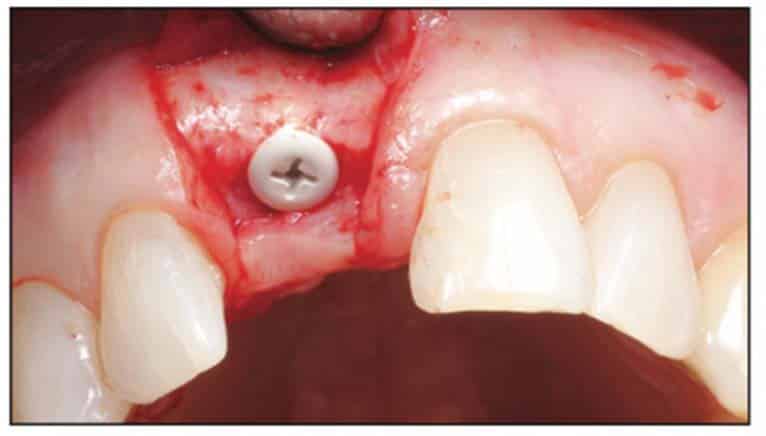
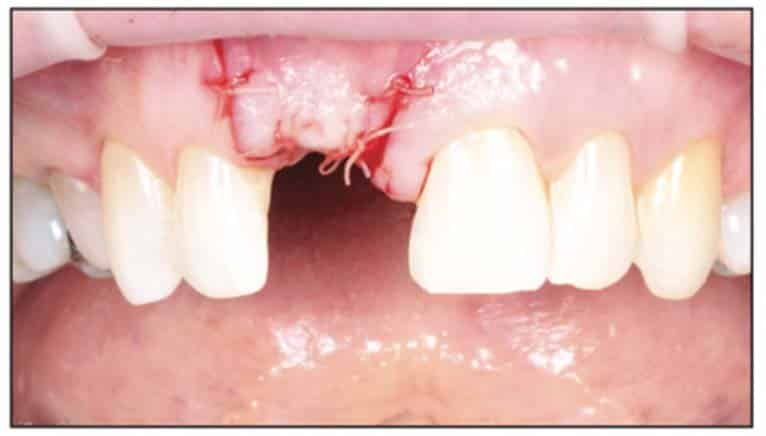
A 2-part reversible screwable zirconium oxide implant (Zeramex XT) made of aluminum-toughened zirconia (ATZ) with a reduced diameter of 3.5 mm and a length of 12 mm was placed under local anesthesia with minimal flap formation. It was then closed with an end cap, and the tissue was repositioned and sutured (Figures 5 to 10).
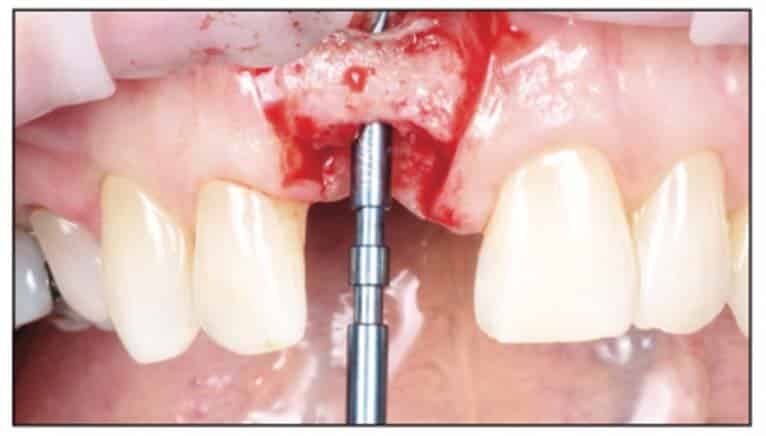
Surgical Phase
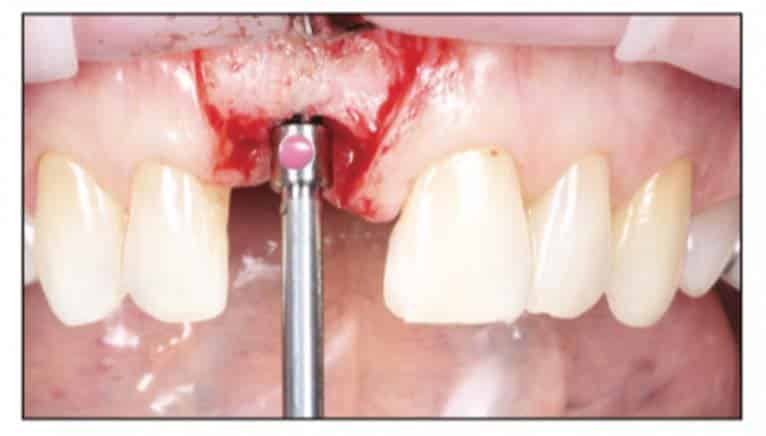
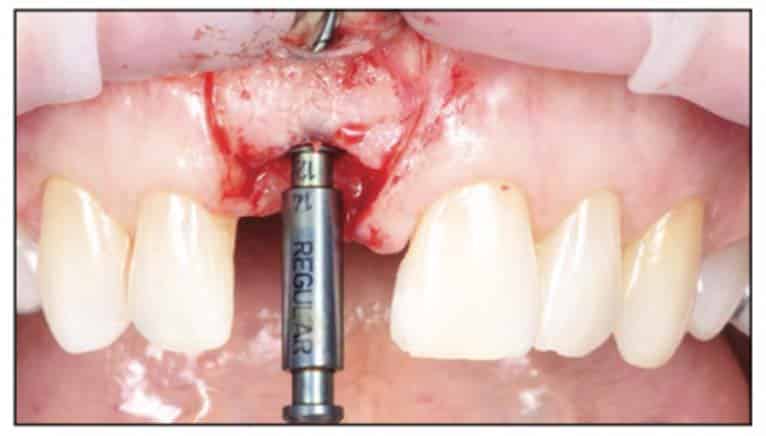
Following the suggested protocol from the manufacturer, the site was prepared for the 2-piece implant placement that was very similar to a titanium variant. It is important to note that the site must be prepared adequately to prevent excessive implant insertion torque and that the use of the drill tap is necessary. One important note when surgically placing the zirconia implant is that the implant is placed 1.6 mm to 0.6 mm supracrestally (Figures 11 to 16). The connection point is to be above the crest of bone. The implant is sandblasted and etched on the body to create a micro-roughened surface with a zone near the neck of the implant that is to not allow for true tissue adhesion.

Soft-Tissue Management and Final Prosthetic Restoration
After the healing phase was completed, the implant was exposed with a diode laser (Brasseler BLU [Brasseler USA]), and a peak Zeramex 3-mm-tall healing cap was placed (Figures 17 and 18). Healing was allowed to continue for 2 weeks (Figures 19 and 20).
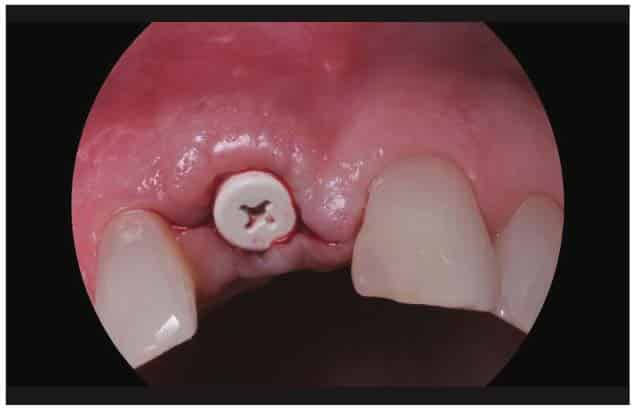
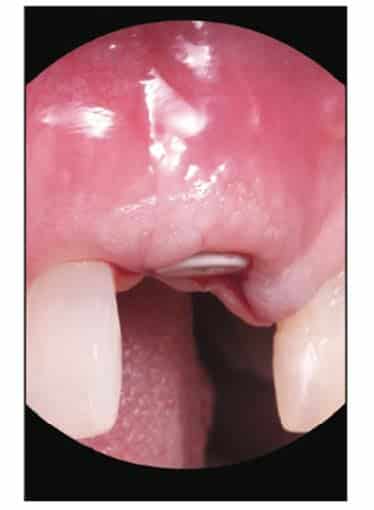
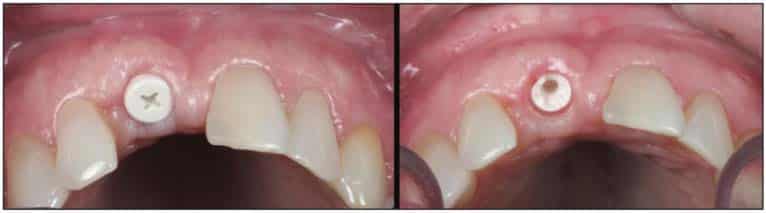
After the soft-tissue healing phase, the gingival profile was evaluated, showed no signs of inflammation, and demonstrated excellent biocompatibility with these materials. A standard restorative process of using a closed tray impression post was planned. After verification with an intraoral PA, a PVS impression was taken (Capture [Glidewell])(Figure 21). This was sent to the lab to fabricate an all-zirconia crown (BruxZir [Glidewell]) (Figure 22).

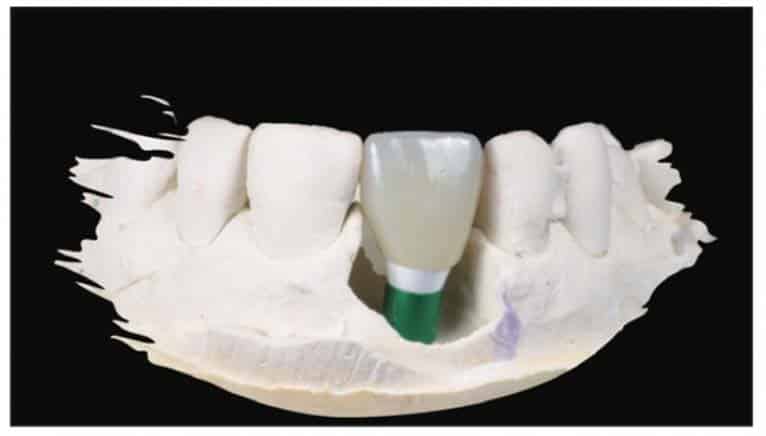
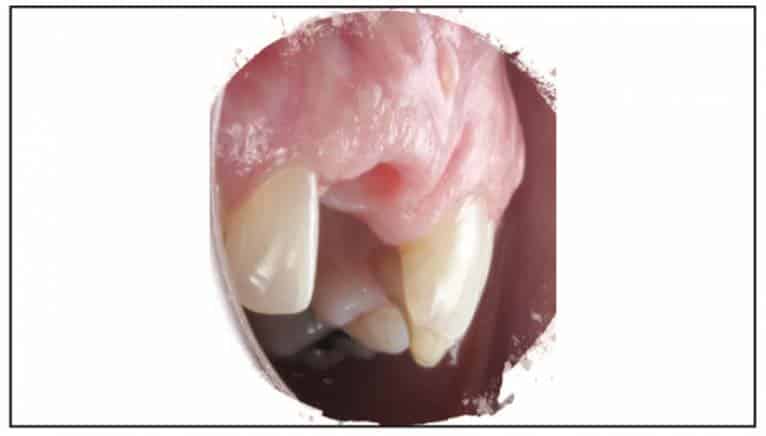
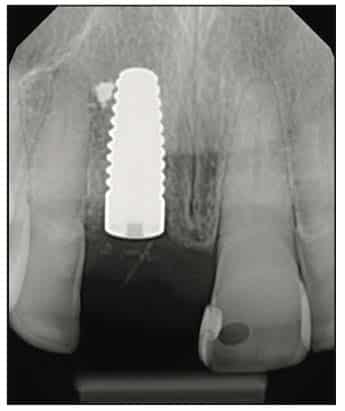
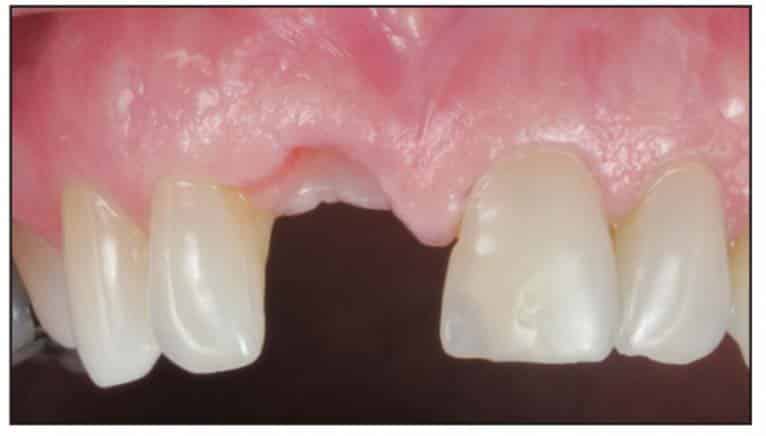
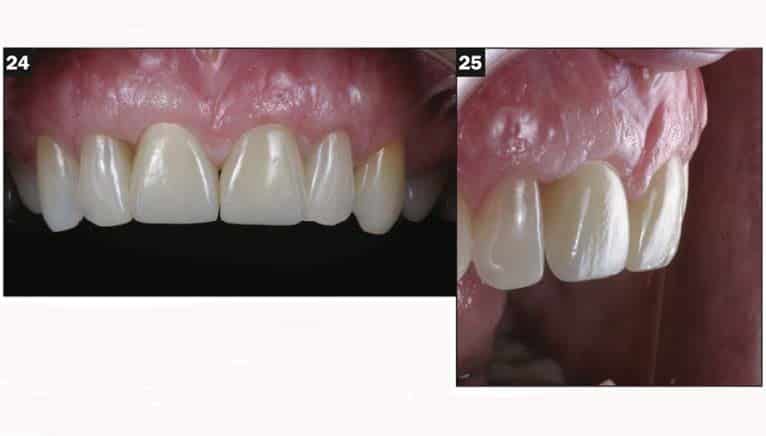
A Zeramex standard abutment also made of ATZ was fixed in the implant with a Vicarbo screw (Zeramex XT). This screw made of carbon fiber longitudinal strands, molded slightly larger than the internal aspect of the implant, allows transmission of the forces of mastication to be absorbed while providing a tight, hermetically sealed connection. The carbon fiber screw affords this complex with almost 2 times the tensile strength of a titanium fixation screw (Figure 23). The final BruxZir crown was tried in and cemented with Infinity cement (DenMat) (Figures 24 and 25).
Register through the form below to download the FULL Case Presentation:
About the Doctor
Dr. Paresh Patel is a graduate of the University of North Carolina at Chapel Hill School of Dentistry and the Medical College of Georgia/AAID MaxiCourse. He is a fellow of the Misch International Implant Institute and a diplomate of the ICOI.
Dr. Patel has placed more than 5,000 implants, has published numerous articles in leading dental journals, and has worked as a lecturer and clinical consultant on dental implants and prosthetics for several companies. He maintains private practices in Lenoir and Mooresville, North Carolina.
Dr. Paresh Patel is a board-certified Doctor of Dental Surgery. He received his doctor training at the prestigious University Of North Carolina. Upon completion of his residency, he went on to pursue advanced training in dental implant surgery.
Early in his career Dr. Patel found his passion in advanced dental procedures including dental implant placement and restoration. This passion and commitment led him to teaching and publishing many of his cases in worldwide dental journals. When Dr. Patel is not running his two office locations in NC, he is traveling around the world teaching 1000’s of dentists the advanced techniques of implant surgery and patient care.
Since 1996, he and his wife, Dr. Manisha Patel, have raised their three children in the greater North Carolina area. In his free time, Dr. Paresh is an avid aviator and enjoys flying regional with friends and family. Dr. Paresh has also developed a passion for photography including unique representations of dentistry as art. Dental Art
Dr. Paresh remains active in both state and local societies. He is honored to have the opportunity to serve and care for patients in this community.
He has a dedicated staff and treats each member like extended family. He leads his staff to care about the patients, share the same practice philosophy, and develop a passion to see growth of our practice.
Dr. Paresh Patel can be reached at:
Office: 828 754 7881 | Office address: 310 Mulberry St SW, Lenior NC, 28645
Contact
Want to know more about Zeramex?
Email us at mfoley@zeramexusa.com with your contact details and we will get in touch with you!
ZERAMEX BROCHURE
Click here to download the Zeramex sales brochure


